Science
 GENES AND ENVIRONMENT
GENES AND ENVIRONMENT

Genes make no sense without consideration of the environment they operate in.
by John McManamy

Genes make no sense without consideration of the environment they operate in.
by John McManamy
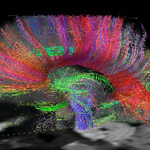
IN 2003, the journal Nature cited a series of studies that essentially changed how we think about mental illness. Most of the studies had to do with a variation in the serotonin transporter gene. As I heard one of the researchers involved in one of the studies, Daniel Weinberger of the NIMH, explain it a few years later this particular gene "impacts on how threatening the environment feels."
One of the end results of this particular gene-environment two-step could be depression. The gene variation itself does not cause depression, so in no way can this be called a depression gene. Moreover, if you look at the gene in terms of pure mechanics, it is just a biological unit that switches on something in the brain.
"If you ever want to know whether your tax dollars are being used for a good purpose," wrote Barbara Oakley in her 2007 Evil Genes, go take a look at the extraordinary work that the National Institute of Mental Health and other National Institutes of Health are doing in digging out the genetic bases of psychiatric illness."
These studies serve as an excellent teaching tool to illustrate the vast complexity of the brain as it attempts to simultaneously grapple with the genetic hand it has been dealt and the environment that turns these genes loose. Key points:
- Our genes and our environment interact. We may not have any choice in changing our genes, but we can often choose to change our environments in a way that lets sleeping genes lie.
- Genes are not deterministic, but they do predispose us to how we react to whatever life may throw our way.
- Stress is the key driving force in mental illness. There are other factors, but stress is invariably complicit. A good deal of mental illness can be summed up as "stress vulnerability disease."
- Diagnostic categories are useful to a point, but malfunctions in serotonin transport have been linked to anxiety, mania, depression, substance abuse, borderline personality disorder, and all manner of things that can go wrong. Likewise, malfunctions in other processes tend to have similar shotgun effects.
- It is more helpful to think of genes switching on and off certain mechanical processes in the brain than "causing" a specific disease. Moreover, these processes work in the context of whole brain systems interacting with other brain systems, which may exacerbate or mitigate the effects of the equivalent of a tap not being able to shut off.
- There are no "good" genes or "bad" genes. Inevitably, there are trade-offs. A gene variation that may predispose you to stress may also protect you from Alzheimer's.
To give us a better understanding of the brain in action, it is useful to look at the impact of other genes. Of all things, Dr Oakley's examples in her book and the ones I cite in various articles on this site are virtually identical. This is no accident, as both Oakley and I draw extensively on the work of Dr Weinberger. In addition to the serotonin transporter gene, Dr Weinberger and his colleagues have been involved with the following:
Brain-Derived-Neurotrophic Factor (BDNF)
This protein is involved in brain cell maintenance and survival and encourages the growth of new neurons and neural connectivity. A "double-val" variation in this gene has been linked to stronger memory. The catch is those with the double-val are prone to more anxiety and moodiness and hostility, possibly due to a magnification of the serotonin transporter glitch. A "double-met," on the other hand may offset the glitch.
Catechol-O-methyltransferase (COMT)
Mechanically, this gene breaks down dopamine and other neurotransmitters. The more slowly you metabolize dopamine (the "met" variation), the smarter you are. The unfortunate "val/vals" may be a bit less intelligent, with a slightly increased risk of schizophrenia, plus risk of antisocial behavior, and hyperactivity. The "val/mets" fall in between. The trade-off? Vals may be able to handle stress better than the mets and be more flexible to change.
Monamine Oxidase A (MAO-A)
This protein breaks down dopamine and other neurotransmitters. Low-functioning MAO-A has been linked to aggressive and antisocial behavior and substance abuse and more. Those with low-efficiency versions of this gene tend to display hyperactive amygdalae (involved in fight-or-flight) and low-responding orbitofrontal and cingulate cortices. In other words, the front end of the brain has problems turning down the alarm signals from the back end of the brain. Impulsive violence may be one result.
A breakthrough 2002 study found that maltreated kids with low-efficiency MAO-A developed significant antisocial problems while the high-efficiency MAO-A kids were better able to weather the storm.
Determinism vs Vulnerability, Changing Our Environment
Dr Oakley makes it abundantly clear that our behaviors are far less governed by free will and reason than our over-sized egos would have us believe. But neither are we helpless bystanders. We have the option of intelligently taking stock of our vulnerabilities and working around them, as well as capitalizing on our assets. We may be stuck with the genetic hands we have been dealt, but we have plenty of options in managing the environments in which these genes operate.
SIGN UP FOR MY FREE EMAIL NEWSLETTER
This came through loud and clear in three lectures I attended at the 2009 American Psychiatric Association annual meeting in San Francisco. We may be shaped by our genes and environment, Robert Cloninger of Washington University (St Louis) acknowledged, but our self-awareness (a uniquely human trait) allows us to modify these influences and move forward with our lives.
Personal change, he said, can happen very rapidly, if you can get the person to recognize their strengths and weaknesses.
According to psychiatric geneticist Kenneth Kendler of the University of Virginia, the environment may neutralize genes. Say, for instance, it is difficult for a kid to obtain alcohol. Then certain alcoholism vulnerability genes have less chance to kick in. Conversely, "our brains have feet." We're not just passive recipients of our environment. Rather, our genes cause us to create the environment around us.
These were points reinforced by Marc Schuckit of UCSD. Dr Schuckit has tracked some 400 offspring of alcoholics and control subjects for more than a quarter century. There are many causes of alcoholism, involving many genes and environmental influences, but Dr Schuckit noticed that when those with alcoholism described their early drinking experiences, they tended to remark how they could drink everyone under the table.
The technical term is "low level of response" (LR). These individuals need to have a lot more drinks to feel the same effect as their peers, and kids drink for effect. Once you get into the pattern of heavy drinking, Dr Shuckit said, then the environment kicks in: You hang out with heavy drinkers; they expect you to engage in heavy drinking.
So, say you know that your 12-year-old has a genetic predisposition to low LR, is there anything you can do about his environment to protect him? Of course there is.
And to bring the conversation back to where we started: If you know that a stressful environment renders you vulnerable to depression, are there things you can do to change your environment, or at least manage it a bit better? Are there tools you can use to handle stress?
Do you see brain science and recovery working together? Please check out the articles in the Recovery section.
See also Psychiatry's Big Bang
Reviewed July 11, 2016
NEW!
Follow me on the road. Check out my New Heart, New Start blog.